| HEAD OF TEAM | : | Astri Nur Istyami, M.T. |
| TEAM MEMBERS | : | Jumadi, Septian Fadillah |
| OFFICIAL ADDRESS | : | Chemical Engineering Program, Labtek X, ITB Ganesha Campus |
| : | anistyami@che.itb.ac.id | |
| EXTENDED ABSTRAct | : |
Introduction
Free fatty acids is an important intermediate product in oleochemical industry. For decades, production of fatty acids from fats and oils are established commercially at high pressure and high temperature, thus needs huge amount of energy. In consequence, some unwanted side reactions are often occur, such as thermal decomposition, polymerization, hydrogenization, and isomerization. Technology of triglyceride hydrolysis can alternatively be established with lipase enzyme utilization, thus the process is also known as lipolysis. This process is potentially more economic than the conventional one because it feasibly operates in mild operating condition and cause less product deterioration. Due to high production cost of microbial lipase, plant is revealed as an interesting alternative source of lipase.
Preliminary experiments showed that frangipani (Plumeria rubra) latex particulate performs highest lipolytic activity compared to other plant lipase observed, including lipase from castor bean, rice bran, and papaya latex. At optimized condition, lipolysis with 1% frangipani latex particulate achieve 74,54% conversion in 10 hours reaction time. In its crude form, frangipani latex contained heterogenous content of protein per gram, resulting in inconsistency of lipolytic degree. On the other hand, immobilized form of lipase is expected to enable the recycling of enzyme.
The focus in this research is immobilization of lipase to enable reusing of plant lipase during lipolysis process and to enhance the consistency of lipolytic degree.
Methods
Methods used in this experiment was adsorption to polypropylene beads, polyethylene beads, rice bran acetone powder, and encapsulation with calcium alginate.
Results
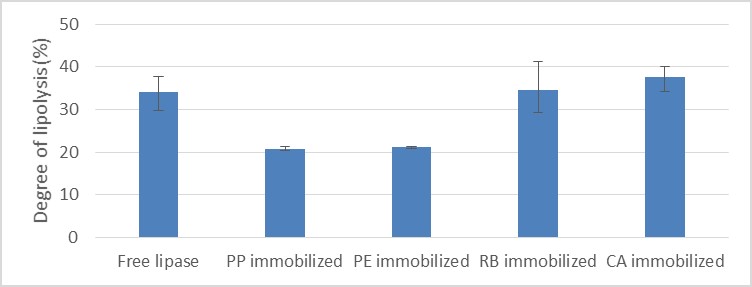 Figure 1. Degree of lipolysis resulted with free lipase, lipase immobilized with polypropylene (PP) beads, polyethylene (PE) beads, rice bran acetone powder (RB), , and calcium alginate (CA)
Figure 1. Degree of lipolysis resulted with free lipase, lipase immobilized with polypropylene (PP) beads, polyethylene (PE) beads, rice bran acetone powder (RB), , and calcium alginate (CA)

Figure 2. Immobilized lipase using polypropylene beads, rice bran acetone powder, polyethylene beads, and calcium alginate
Discussion and Conclusion
Compared to free lipase, immobilized lipase from crude frangipani latex shows lower activity in general. Adsorption of crude lipase on rice bran acetone powder results in high activity retention, but also high standard deviation. Adsorption of crude lipase on polymer beads shows high data consistency, but low activity retention. Encapsulation of crude lipase with calcium alginate results in fair level of activity retention and data consistency.
Adsorption of crude lipase on rice bran acetone powder was inefficient because both of crude lipase and rice bran remained on powder form until reaction ends. Meanwhile, polymer beads did not show significant interactions with crude lipase itself. The most efficient method was encapsulation with calcium alginate, which results in high activity retention compared to free lipase. Recycling of immobilized lipase was not possible on the 2nd cycle.
In general, immobilization of crude frangipani latex was successfully established through encapsulation with calcium alginate. Nevertheless, the frequency of recycling was limited, thus in case of crude frangipani latex lipase utilization, economic feasibility was insignificantly changed. Free lipases are most likely be practical option because the lipases are low-cost and abundantly available.
